Matt Haave
I am a healthcare product, program, and people leader with 15 years experience in a variety of leadership roles. With a portfolio of products that spans scientific equipment, high-volume disposable medical devices, and digital health products, my work blends technical depth, disciplined execution, and people‑first leadership.
"Under his leadership, [Matt’s] teams experienced strong retention, clear development trajectories, and a sense of purpose…"- Davide Ricci, VP US SaMD, Novo Nordisk


People Leadership
I lead by creating clarity, building trust, and empowering teams to do their best work. At Novo Nordisk I managed multiple cross‑functional Data Science, Engineering, and UX teams, fostering psychological safety, strong global collaboration, and a culture of focused, rigorous innovation.
My focus is developing leaders while delivering projects. Through structured coaching, clear expectations, and a healthy cross-functional environment, my teams consistently achieved clinical trial readiness and delivered high‑impact innovation programs, and my direct reports have gone on to succeed in their own leadership roles.
I focus on architecting rigorous, structured engineering processes that ensure quality and compliance while ensuring effort is focused on high-impact tasks.

Product Leadership
I define product vision and strategy that translate clinical and commercial needs into scalable digital health solutions. I’ve led a portfolio of SaMD products from prototype through clinical validation, delivering MVPs that have potential to personalize treatment for hundreds of thousands of patients.
My product management approach prioritizes structured collaboration in highly matrixed organizations. This ensures that user research, engineering, regulatory, clinical, commercial, and data science stay synchronized so that scope creep can be managed and execution is dependable.
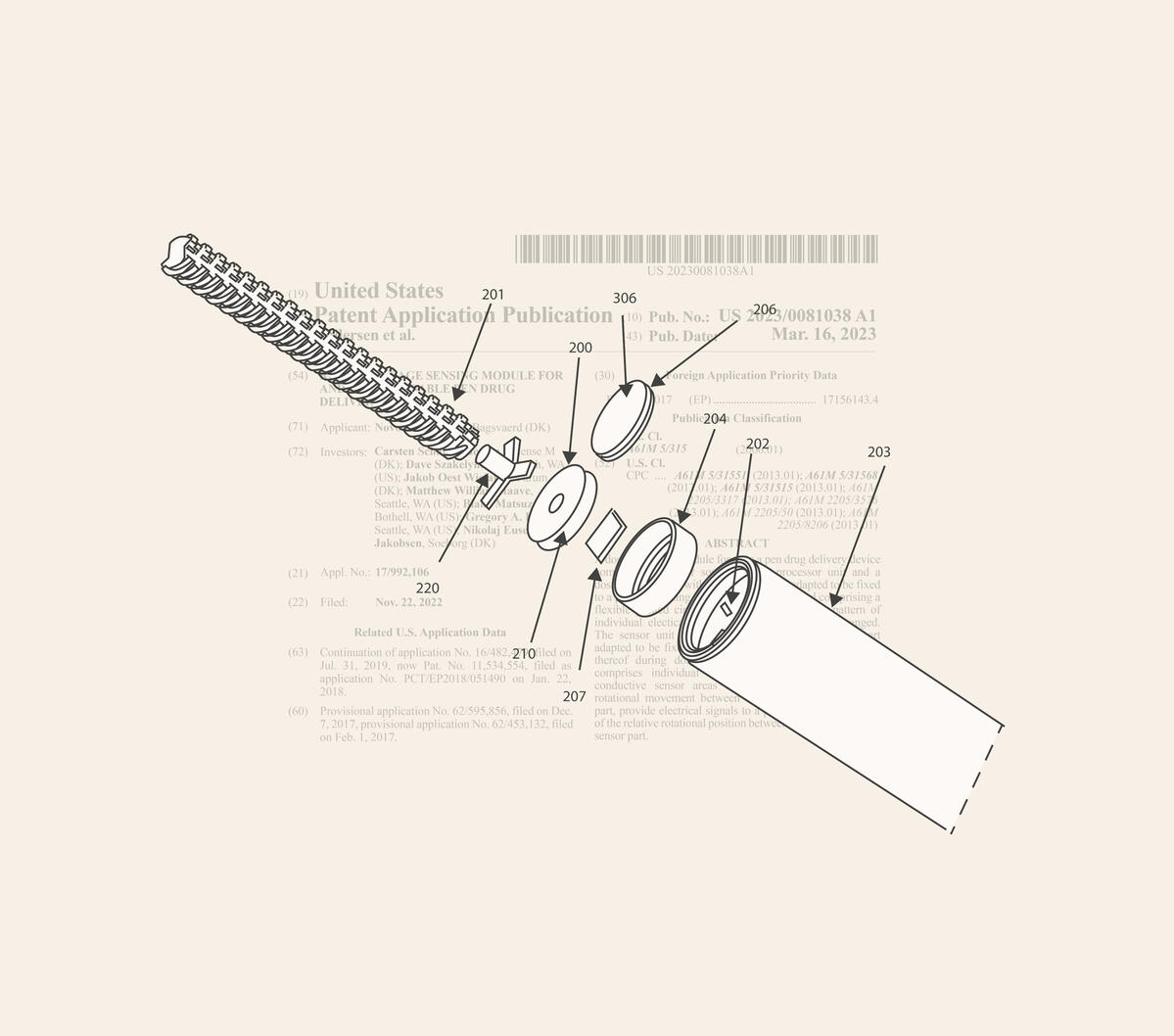
Technical Leadership
My technical leadership is grounded in my background as a physicist and lead engineer on a range of scientific and medical products that have been patented and/or succeeded in clinical testing.
I’ve led the design of high‑volume connected devices (see image) and introduced engineering frameworks that reduced development time across dozens of programs.
I focus on architecting rigorous, structured engineering processes that ensure quality and compliance while ensuring effort is focused on high-impact tasks.

AI & Data Science
I apply AI to solve practical problems in regulated healthcare, from personalization to simulation. My past work includes leading AI‑enabled product initiatives and building RAG‑based tools that support regulated medical device engineering.
I also led data science initiatives that resulted in new medical treatment algorithms and products. LLM-powered products introduce unique regulatory challenges in the healthcare space. By partnering closely with data scientists and clinicians, I ensure models translate into features that are auditable, trustworthy, and aligned with regulatory expectations.

Digital Health & Regulated Development
I bring end‑to‑end experience in SaMD and medical device development, including design controls, risk management, supplier oversight, and clinical trial readiness. I’ve delivered regulated B2B SaaS products into commercialization pipelines and unblocked stalled programs through improved engineering and risk processes.
By bringing an understanding of the technology, processes, regulatory landscape, and industry, I am able to help digital health programs focus and deliver on the most impactful opportunities.
©2026 Matt Haave